Purpose
The Mohr titration method uses the standard solution of silver nitrate, and the silver complexes with the chloride in our sample, react with the chromate added to the sample to form silver chromate. The amount of silver reacted with chloride is used to determine how much sodium was in our sample.
The Mohr titration method uses the standard solution of silver nitrate, and the silver complexes with the chloride in our sample, react with the chromate added to the sample to form silver chromate. The amount of silver reacted with chloride is used to determine how much sodium was in our sample.
|
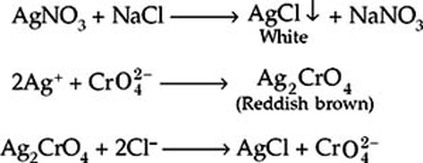
To the right is the chemical equation as to how sodium chloride reacts with silver nitrate and then with silver chromate to form silver chloride and chromate.
After the silver nitrate has complexed with all of the chloride contained in the sample, the silver reacts with the chromate in the sample to form an reddish brown color solid, which is the silver chromate.
|
|
Procedure We prepared the primary standard solution in duplicate. We first transferred 400 mL of 0.1 AgNO3 solution into a bottle and covered the bottle with aluminum to reduce the exposure to light. We then filled the burette with AgNO3 solution. To prepare the primary standard solution, we weighed about 100 mg KCl into three 125 mL Erlenmeyer flasks, added 2 drops of K2CrO4 solution and 25 mL of water. We placed a stir bar in the flask, and placed under stir plate. We titrated KCl solutions with the AgNO3 until the color of the solution changed to permanent pale, pink-orange color. We recorded the volume of AgNO3 used for titration, and calculated the molarity of AgNO3. |
|
For our sports drink sample, we placed 5 mL of our sample in duplicate into 250 mL beakers, and added 95 mL of boiling water into each beaker. We stirred the mixture vigorously using stir bar and stir plate for 30 seconds, waited 1 minute, and stirred again for 30 seconds. 50 mL of each solution were transferred into 250 mL Erlenmeyer flasks. 1 mL of potassium chromate indicator was added to each 50 mL prepared sample. Each sample was titrated with the standardized 0.1 M AgNO3 until the color of the solution persisted as pale red-brown color for 30 seconds. We then recorded the volume of titrant used.
|
Results and Conclusion
% chloride= 0.0013
% sodium chloride= 0.002166
% sodium= 0.000866
% error= 61.58 %
Average molarity of AgNO3 solution= 0.09256 M AgNO3
In this experiment, we determined the amount of sodium chloride and chloride in our sample, and used the values to calculate the amount of sodium. Our percent sodium was about 0.000866, and we got 61.58% error. The possibility for such high error is that the silver nitrate may have been exposed to light for quite some time prior to titration, causing a reaction to form silver oxide and nitric acid. This may have affected the binding of silver with chloride, thus affecting the binding of chromate. Another possibility is the temperature of the boiling water. When we added the 95 mL of boiling water into our 5 mL sports drink sample, the temperature of the water cooled down from boiling to warm, which may have affected the interaction between silver and chloride in later steps of the protocol. Using AgNO3 exposed to less light, using boiling water, and adjusting the lab settings to superlative condition may help us collect better results and reduce the percent error.
% chloride= 0.0013
% sodium chloride= 0.002166
% sodium= 0.000866
% error= 61.58 %
Average molarity of AgNO3 solution= 0.09256 M AgNO3
In this experiment, we determined the amount of sodium chloride and chloride in our sample, and used the values to calculate the amount of sodium. Our percent sodium was about 0.000866, and we got 61.58% error. The possibility for such high error is that the silver nitrate may have been exposed to light for quite some time prior to titration, causing a reaction to form silver oxide and nitric acid. This may have affected the binding of silver with chloride, thus affecting the binding of chromate. Another possibility is the temperature of the boiling water. When we added the 95 mL of boiling water into our 5 mL sports drink sample, the temperature of the water cooled down from boiling to warm, which may have affected the interaction between silver and chloride in later steps of the protocol. Using AgNO3 exposed to less light, using boiling water, and adjusting the lab settings to superlative condition may help us collect better results and reduce the percent error.