Purpose
High Performance Liquid Chromatography (HPLC) is an advanced form of liquid chromatography, which allows solvent to pass through the column through force high pressure of up to 400 atmospheres. Compared to the traditional column chromatography, HPLC is faster, and allows for better separation of the components of the mixture through the use of smaller particle sizes for the column packing material, providing greater surface area for interactions between the mobile and stationary phases of the molecules passing through the column.
Components of the HPLC System:
High Performance Liquid Chromatography (HPLC) is an advanced form of liquid chromatography, which allows solvent to pass through the column through force high pressure of up to 400 atmospheres. Compared to the traditional column chromatography, HPLC is faster, and allows for better separation of the components of the mixture through the use of smaller particle sizes for the column packing material, providing greater surface area for interactions between the mobile and stationary phases of the molecules passing through the column.
Components of the HPLC System:
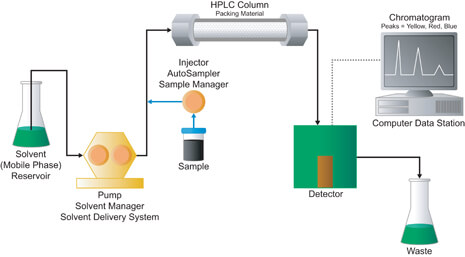
Components of the HPLC system are shown in the diagram above. A reservoir contains the solvent (mobile phase), and a system of two high pressure pumps. The two pumps enable the use of two different solvents, allowing for gradual switch from one solvent composition to another. The pumps are also used to generate and measure a specific flow rate of the mobile phase, in mL/min. The injector allows us to inject the sample using the Hamilton Syringe into the continuously flowing mobile phase stream that carries the sample into the HPLC column. The mobile phase is then sent to waste, and the data is sent to the computer. The detector is connected to the computer data station, which records the electrical signal needed to generate the chromatogram and identify the concentration of the sample constituents.
Here is a quick and simple animation on how the HPLC works:
Procedure
We mixed 5 mL of our juice sample with 5 mL of 5% metaphosphoric acid, and centrifuged for 10 minutes at 5°C. We treated a disposable C18 Sep-Pak cartridge with 3 mL of methanol followed by 5 mL of water. We then loaded 5 mL of the centrifugate onto the c18, and we discarded the first 2 mL of eluate, and collected the next 2 mL. The 2mL were filtered through 0.45 um Millipore filter before injection. We repeated these steps for all of the juice samples (chilled, room temperature, and boiled)
20 uL of aliquot of one of our sample were analyzed through the analytical column, at 245 nm because the HPLC took a very long time to be analyzed.
Result and Conclusions
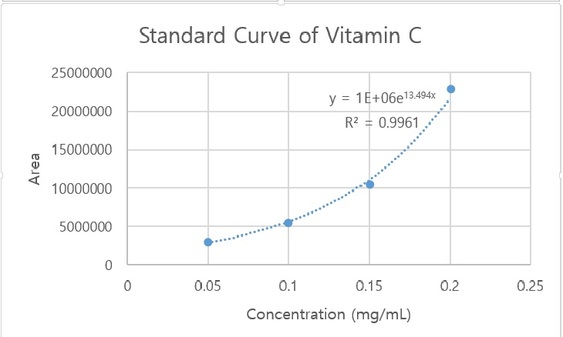
The standard curve of vitamin C (ascorbic acid) is plotted below. The calculations for ascorbic acid concentrations were determined using this standard curve model, which had a strong correlation coefficient of 0.9961.
The sample analysis using both the standard curve and the HPLC were very similar, as we got a concentration of 0.1960 mg/mL from the standard curve and 0.2034 mg/mL from the HPLC. We have a 3.78 % error, indicating that the accuracy of HPLC was very high. However, in order to reduce our percent error even more, we would consider duplicating or running several trials in measuring the concentration of ascorbic acid by HPLC. Also, as we are running several trials for accurate results, reducing the exposure of oxygen and air to our samples would lessen the process of oxidation and lead to better results.
|
Fun Analogy of the Day
THE RUBBER DUCK RACE A rubber duck race is held at your local area, and brings about a million of its mini-sized rubber duck friends labeled with a number to participate in a race. They are then set out to flow in the river and race against each other to see which duck can travel the fastest to the endpoint. Some small duckies might eventually get caught off by rocks, dead branches, or be brought up to the shore, while other duckies may be successful in swimming to the endpoint of the river. |
|
Now let's compare rubber ducks to HPLC!
The million rubber ducks are like our sample mixture we want to analyze. The river is our column, and the water is our solvent. Some ducks are collected in certain areas of the river due to sinking under water or possible rocks or dead branches hindering the movement of the ducks; this is the same as the components of the sample mixture having different characteristics that determines the rate at which they can reach the bottom of the column (the endpoint of the river). |