Purpose
Calorimetry is the measurement of quantities of heat. It involves the use of temperature and energy units but is not a measurement of temperature. The value of a calorie describes the heat of combustion or the number of heats units release by a sample that has been burned with oxygen in an enclosure of constant volume. The bomb calorimeter measures the amount of heat release by the combusiton of carbon , hydrogen and oxygen used to form carbon dioxide and water. The results of the reaction that takes place in a bomb calorimeter are expressed in Calories. One Calorie= 4.1868 Joules and this is equal to the amount of energy needed to raise the temperature of one gram of water by one degree Celsius at a pressure of one ATM.Thus, the purpose of this lab is to become familiar with the bomb calorimeter and to use it to identify the caloric density of our product.
Calorimetry is the measurement of quantities of heat. It involves the use of temperature and energy units but is not a measurement of temperature. The value of a calorie describes the heat of combustion or the number of heats units release by a sample that has been burned with oxygen in an enclosure of constant volume. The bomb calorimeter measures the amount of heat release by the combusiton of carbon , hydrogen and oxygen used to form carbon dioxide and water. The results of the reaction that takes place in a bomb calorimeter are expressed in Calories. One Calorie= 4.1868 Joules and this is equal to the amount of energy needed to raise the temperature of one gram of water by one degree Celsius at a pressure of one ATM.Thus, the purpose of this lab is to become familiar with the bomb calorimeter and to use it to identify the caloric density of our product.
Procedure
The sample was retrieved from the freeze dryer and weighed to get the dry wet. The sample was then milled to get a granular texture suitable for pellet formation. After the milling process, the sample was divided into four samples, three will be used in future experiments. Using one of the four samples two new samples were created and weighed (about.6 grams). The new samples were then pelleted (using a pellet machine) and weighed afterwards. Each pellet was placed in bomb calorimeter, he bomb calorimeter calculated the calories/gram, at a given sample size.
The sample was retrieved from the freeze dryer and weighed to get the dry wet. The sample was then milled to get a granular texture suitable for pellet formation. After the milling process, the sample was divided into four samples, three will be used in future experiments. Using one of the four samples two new samples were created and weighed (about.6 grams). The new samples were then pelleted (using a pellet machine) and weighed afterwards. Each pellet was placed in bomb calorimeter, he bomb calorimeter calculated the calories/gram, at a given sample size.
Results and Conclusion
Wet weight= 52 grams
Dry weight= 49 grams
Wet weight= 52 grams
Dry weight= 49 grams
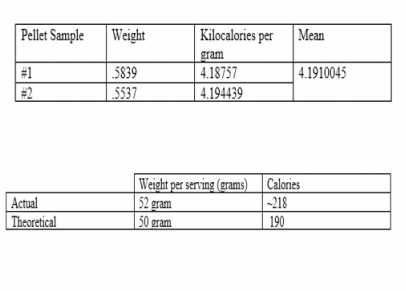
The process of obtaining the proper caloric density of a product is a time consuming and delicate process, especially when it comes to the use of a calorimeter. Inputting the proper weight of the sample, making sure the test sample is in the form of a compact pellet, having enough oxygen, along with other factors could affect not only the operation of the calorimeter but the final results as well. Our average kcal per gram of our sample was higher than the theoretical by about .39 grams, this creates an overall caloric difference of ~ 8.4 when actual kcal/g is multiplied by the theoretical weight. This difference can possible be attributed to the difference in bomb calorimeter, though theoretically the process is the same, the specifics of it may vary. It can be predicted that the other nutritional contents of the product will also differ and will not adhere conservatively to the values of the nutritional label.