Purpose
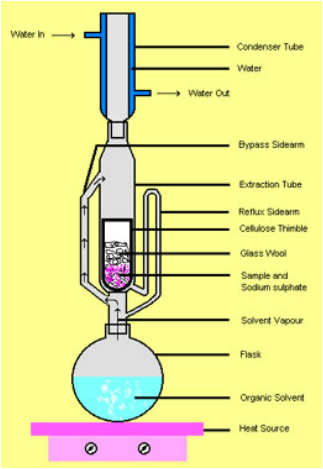
Lipids can be extracted using several methods. Non-polar solvents are especially important in fat extraction given that lipids are non-polar molecules. The three main method types employed for lipid extraction are the continuous, semi-continuous, and discontinuous methods. The Soxhlet method is an example of a semi-continuous method. However, the method chosen depends on several factors including the types of lipids in the sample, the non-lipid composition of the sample, cost, time constraints, and desired accuracy. There are several key steps that comprise the Soxhlet method. First, a non-polar solvent is heated and it rises above the sample through a bypass sidearm.Consequently, once it reaches a water-cooled condenser, is re-enters the liquid phase and falls onto the sample that is contained in the cellulose thimble. At this point, the sample is emulsified with non polar solvent which will extract the lipid molecules from the food matrix. A siphon is used to direct solvent with lipid material back to the round bottom flask. Once the extraction is done, the volatile solvent from the flask is evaporated so that only lipid will be left in the flask.
Procedure
One gram of our lyophilized Pop-Tart sample was weighed on weighing boat. The thimbles were filled placed into the center component of the extraction tube. The non-polar solvent that we used was petroleum ether. The extraction was allowed to run for 1 hour. After extraction, the liquid was emptied into preweighed scintillation vials and the petroleum ether was allowed to evaporate in a water bath in the presence of inert nitrogen gas. Vials were reweighed once the ether evaporated and the amount of lipid was determined.
Lipids can be extracted using several methods. Non-polar solvents are especially important in fat extraction given that lipids are non-polar molecules. The three main method types employed for lipid extraction are the continuous, semi-continuous, and discontinuous methods. The Soxhlet method is an example of a semi-continuous method. However, the method chosen depends on several factors including the types of lipids in the sample, the non-lipid composition of the sample, cost, time constraints, and desired accuracy. There are several key steps that comprise the Soxhlet method. First, a non-polar solvent is heated and it rises above the sample through a bypass sidearm.Consequently, once it reaches a water-cooled condenser, is re-enters the liquid phase and falls onto the sample that is contained in the cellulose thimble. At this point, the sample is emulsified with non polar solvent which will extract the lipid molecules from the food matrix. A siphon is used to direct solvent with lipid material back to the round bottom flask. Once the extraction is done, the volatile solvent from the flask is evaporated so that only lipid will be left in the flask.
Procedure
One gram of our lyophilized Pop-Tart sample was weighed on weighing boat. The thimbles were filled placed into the center component of the extraction tube. The non-polar solvent that we used was petroleum ether. The extraction was allowed to run for 1 hour. After extraction, the liquid was emptied into preweighed scintillation vials and the petroleum ether was allowed to evaporate in a water bath in the presence of inert nitrogen gas. Vials were reweighed once the ether evaporated and the amount of lipid was determined.
Results and Conclusion
The lipid extract that we were able to acquire was a golden brown color that boasted a caramel aroma. Our data can be observed below.
Sample Mass of Sample (g) Mass of Lipid (g)
1 1.0060 0.0836
2 1.0030 0.0732
Calculations:
Sample 1: (lipids g)/(dry sample g) .0836g/1.0060g = .083101 g of lipid/g of sample
Sample 2: (lipids g)/(dry sample g) .0732g/1.0030g = 0.07298 g of lipid/g of sample
Average: 0.078004 g lipid/g of sample
Gram of lipid in a serving (50 g): 0.078004 x 50 g = 3.90 g of lipid
Nutritional Label for lipid: 4.5 g
% Error: 13.3 %
Gram of lipid in a serving (50 g): 0.078004 x 50 g = 3.90 g of lipid
Nutritional Label for lipid: 4.5 g
% Error: 13.3 %
The lipid extraction experiment was a way to quantify and compare label values to experimental values for fat. This experiment showed significant (13.3%) difference between label values and experimental values. These findings indicate that there may have been significant source of error in the process. One such source of error could have been spilling of the solvent and extract from the round bottom flask. One lab member did touch with bare hands the vial. However, the lipids from the hand should have been added to lipid mass instead of decreasing lipid mass. More investigations should be done to find the problems and discrepancies.