Purpose
Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis (SDS-PAGE) is a unique analytical tool used to separate proteins in a solution along a polyacrylamide gel. It is an excellent diagnostic for assessing protein purity. Moreover, the molecular weight of proteins in the mixture can be determined by comparing a protein band to its relative location along a protein ladder standard.
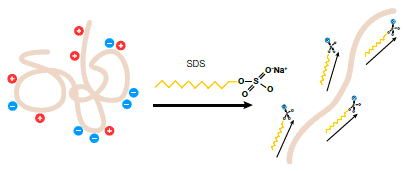
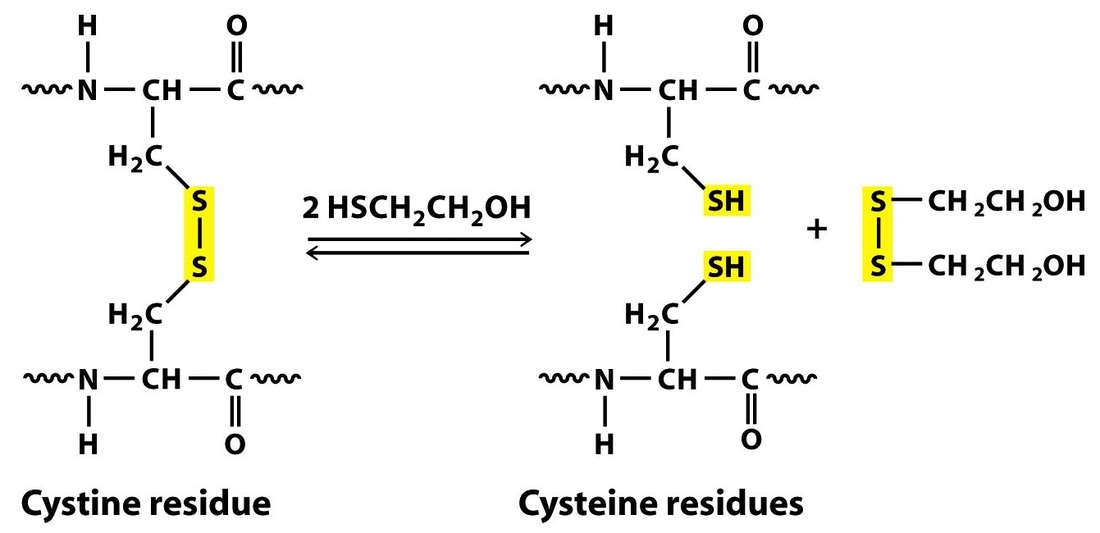
The theory behind this assay is rather simple. First, a solution of proteins is mixed with a detergent named Sodium Dodecyl Sulfate (SDS). What this accomplishes is that proteins become denatured/linear and proportionally coated with the SDS molecule that gives them uniform mass to charge ratio. The proteins may be further mixed with beta-mercaptoethanol if they are known to contain disulfide (cystine) bonds. The samples are then run on a polyacrylamide gel in the presence of an electric field, with the positive electrode near the bottom of the gel. Since the proteins are coated with negative charge from the SDS, they travel down the gel towards the positive electrode. However, their rate of migration down the gel is determined by the relative size of the proteins. Small proteins migrate at a rapid pace compared to larger proteins which are much slower to make their way through the pores of the polyacrylamide gel. Furthermore, the protein bands from a SDS-PAGE gel can be transferred to a nitrocellulose or nylon membrane and then tagged with primary and secondary antibodies in order to perform a western blot. This assay would be useful if we wanted to prove the identity of each band on our gel.
Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis (SDS-PAGE) is a unique analytical tool used to separate proteins in a solution along a polyacrylamide gel. It is an excellent diagnostic for assessing protein purity. Moreover, the molecular weight of proteins in the mixture can be determined by comparing a protein band to its relative location along a protein ladder standard.
The theory behind this assay is rather simple. First, a solution of proteins is mixed with a detergent named Sodium Dodecyl Sulfate (SDS). What this accomplishes is that proteins become denatured/linear and proportionally coated with the SDS molecule that gives them uniform mass to charge ratio. The proteins may be further mixed with beta-mercaptoethanol if they are known to contain disulfide (cystine) bonds. The samples are then run on a polyacrylamide gel in the presence of an electric field, with the positive electrode near the bottom of the gel. Since the proteins are coated with negative charge from the SDS, they travel down the gel towards the positive electrode. However, their rate of migration down the gel is determined by the relative size of the proteins. Small proteins migrate at a rapid pace compared to larger proteins which are much slower to make their way through the pores of the polyacrylamide gel. Furthermore, the protein bands from a SDS-PAGE gel can be transferred to a nitrocellulose or nylon membrane and then tagged with primary and secondary antibodies in order to perform a western blot. This assay would be useful if we wanted to prove the identity of each band on our gel.
Procedure
The experiment that we performed consisted of analysis of two different protein samples. One of these was a skim milk sample and the other was one of just casein protein.
The experiment that we performed consisted of analysis of two different protein samples. One of these was a skim milk sample and the other was one of just casein protein.
These were
mixed with running buffer that contained SDS. Beta-mercaptoethanol was then added to eliminate any disulfide bonds in these proteins. Subsequently, only 20 microliters of
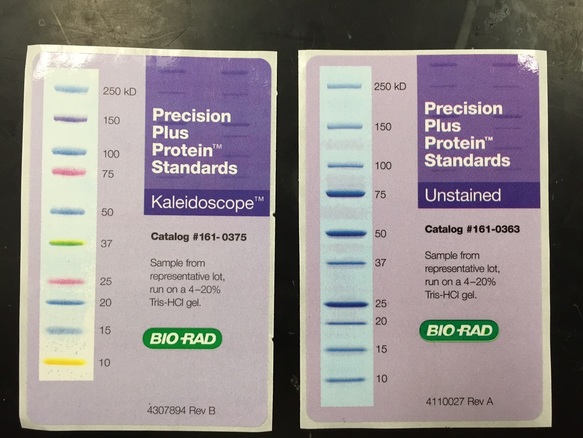
sample were loaded into the wells of the 12 % polyacrylamide gel along with two ladder standard samples.
Once all of the samples were loaded, we introduced an electric field by connecting the gel box to a power supply. The gel was run for fifty minutes and subsequently stained with coomassie blue and then rinsed with destain solution in order to visualize the bands of protein. The video below details the steps in running a SDS-PAGE gel:
Results and Conclusions
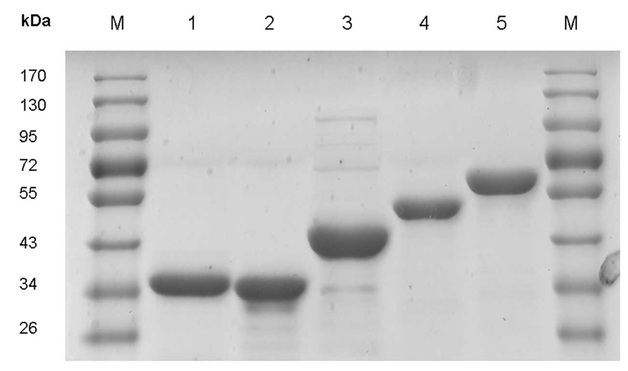
We were not able to attain good results in this exercise. However, we can discuss the results that we should have observed. Theoretically, we should have been able to see a number of bands in the skim milk lane since there are a variety of proteins in milk. On the contrary, we should have only seen one band in the casein lane. Had we seen other bands, it would have indicated that the casein solution was not pure. Also, we could have been able to identify the band for casein in the skim milk lane since we had these two lanes next to each other. Below is a picture of a gel with nicely resolved protein bands.